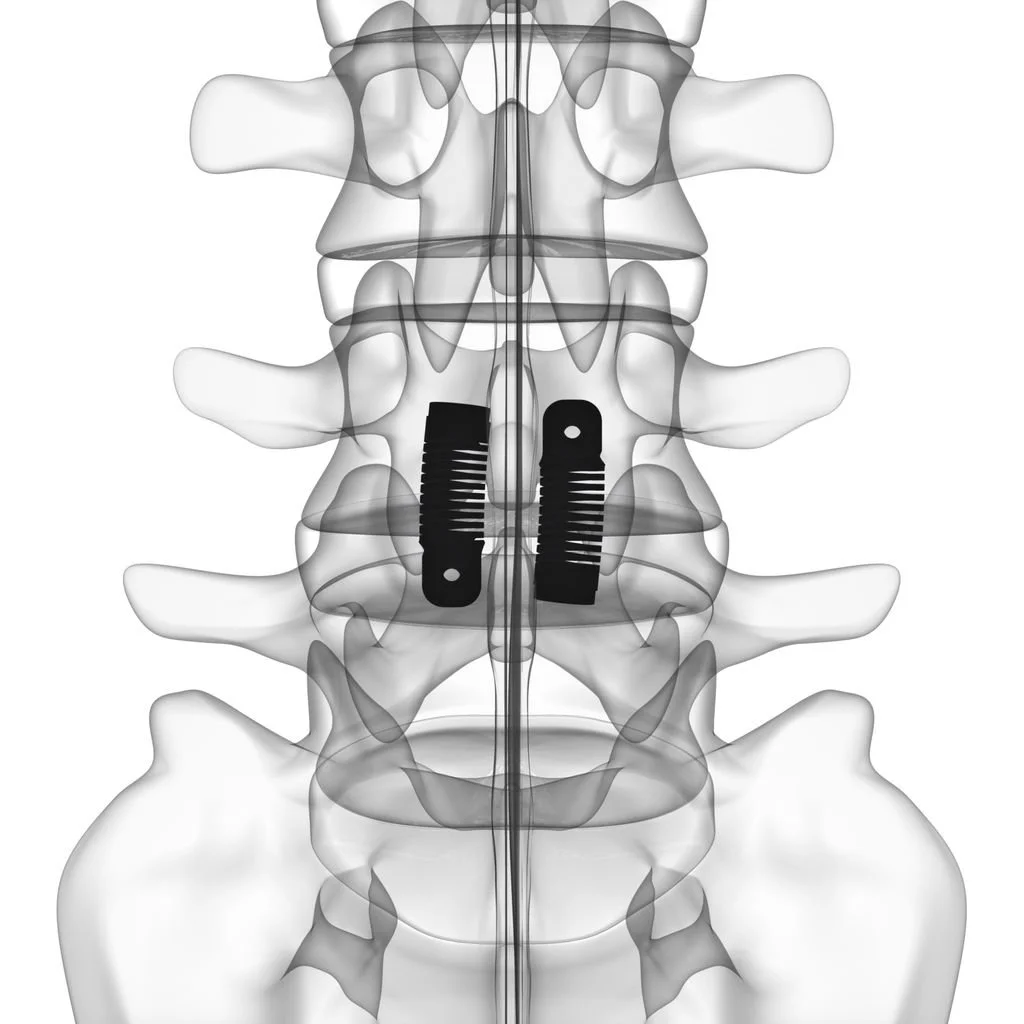
The U.S. Food and Drug Administration (FDA) has granted Premarket Approval (PMA) to the LimiFlex Dynamic Sagittal Tether, a motion-preserving system for the treatment of lumbar spinal stenosis associated with Grade I degenerative spondylolisthesis following decompression.
Read MoreA recently published paper in The Open Orthopaedics Journal compared outcomes for degenerative spondylolisthesis surgery using decompression and Empirical Spine’s LimiFlex Dynamic Sagittal Tether (DST) stabilization versus transforaminal lumbar interbody fusion (TLIF). The interim results of the FDA IDE trial showed both approaches led to substantial improvements in pain and function within the initial postoperative period, but the DST group had shorter hospital stays, earlier return to work, and higher patient-reported activity levels three months post-surgery.
Read MoreEmpirical Spine, Inc., a medical device company creating a new class of spinal implant, recently completed the final step in the U.S. Food & Drug Administration (FDA) submission process for the LimiFlex Dynamic Sagittal Tether (DST). The Premarket Approval (PMA) submission included Module III, with data and analysis of the two-year results from the pivotal Investigational Device Exemption (IDE) clinical trial comparing LimiFlex DST stabilization versus fusion surgery for degenerative spondylolisthesis (degen spondy) patients with spinal stenosis.
Read MoreEmpirical Spine, Inc., a medical device company creating a new class of spinal implant that works in parallel with the natural structures of the spine to restore functionality and optimize quality of life, will highlight its LimiFlex Dynamic Sagittal Tether™ (DST) at the North American Spine Society (NASS) Annual Meeting (Oct. 12 – 15 in Chicago). William Lavelle, MD, Professor of Orthopedic Surgery, Upstate University Hospital, and IDE study investigator, will present interim findings from the US Food & Drug Administration (FDA) LimiFlex DST Investigational Device Exemption (IDE) clinical trial.
Read MoreEmpirical Spine, Inc., a medical device company creating a new class of spinal implant that works in parallel with the natural structures of the spine to restore functionality and optimize quality of life, recently had its Premarket Approval (PMA) Module II accepted and closed by the US Food & Drug Administration (FDA), an important milestone in the commercial approval process for its LimiFlex Dynamic Sagittal Tether™ (DST).
Read MoreEmpirical Spine, Inc., a medical device company creating a new class of spinal implant that works in parallel with the natural structures of the spine to restore functionality and optimize quality of life, recently submitted Premarket Approval (PMA) Module II in the US Food & Drug Administration (FDA) approval process for its LimiFlex Dynamic Sagittal Tether™ (DST).
Read MoreBecker’s Spine Review recognized Todd Alamin, MD as a "Spine Surgeon to Know." Dr. Alamin is a professor of orthopedic surgery and specializes in minimally invasive spine surgery. He is Co-Founder and Medical Director of Empirical Spine, and he is the original inventor of the LimiFlex® device.
Read MoreEmpirical Spine, Inc., a medical device company creating a new class of spinal implant that works in parallel with the natural structures of the spine to restore functionality and optimize quality of life, achieved several clinical, reimbursement and regulatory milestones in the past 12 months that are moving its LimiFlex™ Dynamic Sagittal Tether™ (DST) closer to market in the US. The company also recently initiated several key pre-commercial/market readiness projects.
Read MoreEmpirical Spine, Inc., a Silicon-Valley based medical technology company, announced today that data from the IDE study of the company’s LimiFlex Tension Band for
patients suffering from degenerative spondylolisthesis with lumbar spinal stenosis, was presented at the
Society for Minimally Invasive Spine Surgery (SMISS) Annual Forum 2021. The SMISS Annual Forum took
place from October 28 – 30, 2021 in Las Vegas, Nevada.